
Dr.Ergo® L-Ergothioneine Bulk Powder
−−−Backed by US FDA-recognized GRAS status and supported by two human clinical trials for healthy longevity
Dr.Ergo® L-Ergothioneine is another premium ingredient we offer for supporting healthy longevity. It is produced in a facility certified by NSF cGMP and HACCP, utilizing licensed full enzymatic natural fermentation technology developed by the Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences.
Dr.Ergo® L-Ergothioneine has been granted US FDA GRAS status—only the second ingredient of its kind—and is also authorized as a Novel Food in the EU. Its safety and efficacy have been confirmed through two human clinical trials.
Professor Barry Halliwell, a leading expert on antioxidants and healthy longevity, stated at 2025 Vancouver Ergothioneine Forum that intake of L-Ergothioneine is low among Americans and many other Western populations, which may contribute to poorer healthspan and lifespan. Our Dr.Ergo® L-Ergothioneine is well-positioned to help address this deficiency.


COA of Dr.Ergo® L-Ergothioneine
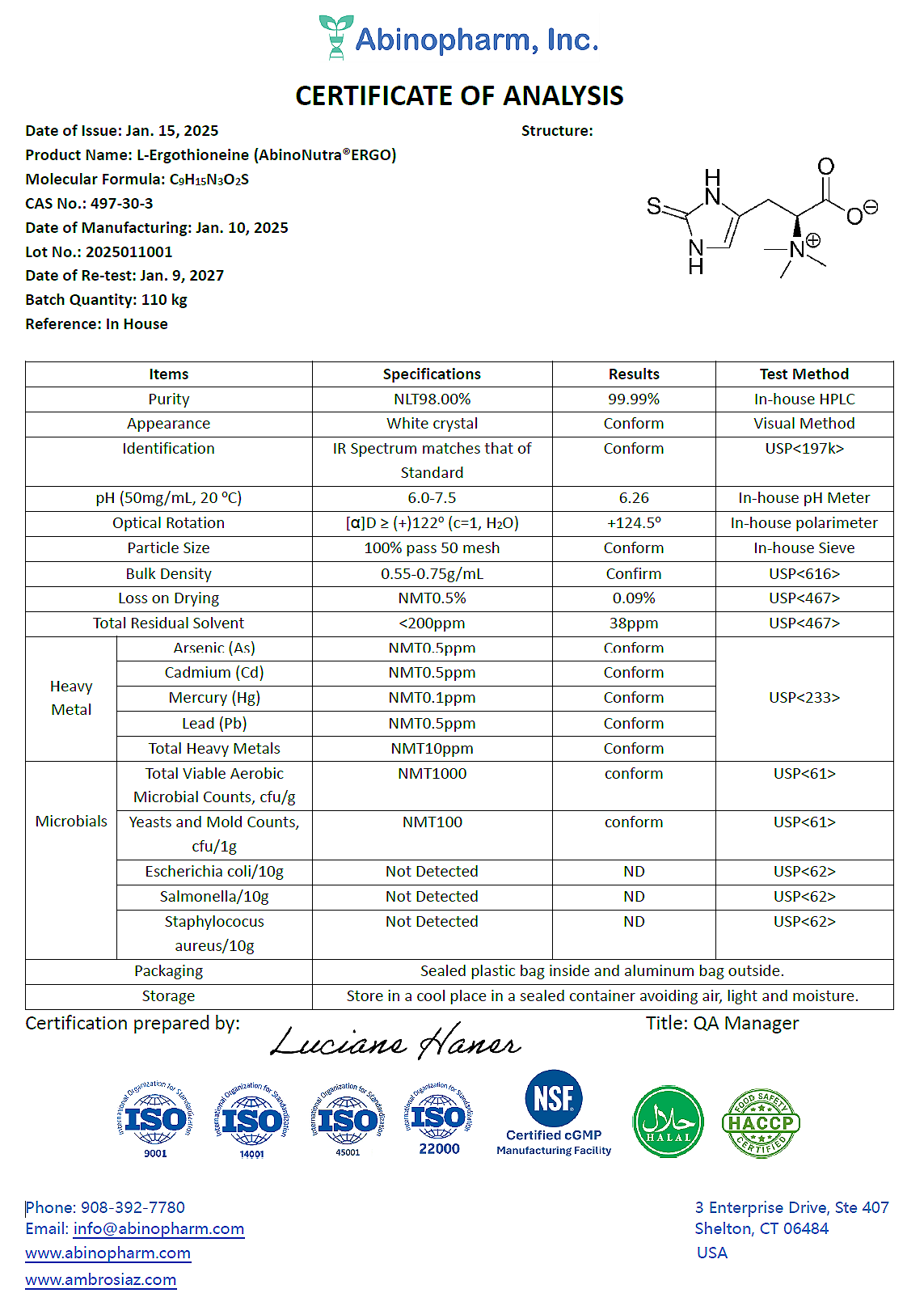
Dr.Ergo® L‑Ergothioneine quality specifications are established in accordance with ICH guidelines for pharmaceutical‑grade materials. Samples from each production batch are tested using United States Pharmacopeia (USP) methods and validated in‑house analytical procedures. A representative Certificate of Analysis (CoA) for a typical batch is provided for reference.
Each CoA lists all tested parameters, including:
Chemical purity
Optical purity (optical rotation)
Identification
Residual solvents
Physical properties (particle size, bulk density, etc.)
Heavy metals
Microbial limits
Batch test results must meet or exceed the predefined quality specifications before the product is released for commercial sale.


